Abstract
Objective: To compare obstetric characteristics and pregnancy outcomes of paztients following two vs. three or more primary recurrent pregnancy losses (RPL).
Study design: A retrospective cohort study including 168 patients with primary RPL followed by subsequent (index) pregnancy, 124 patients with three or more consecutive RPL, and 60 patients with two consecutive RPL was performed. All patients were evaluated and treated in the RPL Clinic in the Soroka University Medical Center.
Results: Obstetric characteristics of the study groups were similar. Women with three or more RPL compared to women with two RPL had a higher rate of abnormal thyroid stimulating hormone (TSH) levels (16.3 vs. 2.6 % ; P = 0.033), higher rates of spontaneous pregnancy (91.7 vs. 77.4 % ; P < 0.05), and higher rates of Clexane treatment (40.3% vs. 18.6 % ; P = 0.016). In the index pregnancy, live birth rate was not statistically different between the two groups (81.1% in the two-RPL groups vs. 70.6 % in the three-RPL group) nor was neonatal mortality.
Conclusions: Patients with two RPL and three RPL had very similar obstetric characteristics and evaluation test results. Differences in index pregnancy outcomes were not statistically signifi cant. Therefore, evaluation in primary RPL is recommended after two RPL.
Introduction
Recurrent pregnancy loss (RPL) also referred to as recurrent miscarriage or by its original term habitual abortion, is historically defi ned as three consecutive pregnancy losses before 20 weeks from the last menstrual period [15] . Based on the incidence of sporadic pregnancy loss, the incidence of RPL should be approximately one in 300 pregnancies. However, epidemiologic studies have revealed that 1 – 2 % of women experience RPL [22] . Recently, the defi nition has been changed to two consecutive losses [1, 23, 24] and, as a consequence, the rate of RPL increased from 1 to 5 % . The new defi nition was based on developments in medical fi elds, such as new laboratory tests including antiphospholipid antibodies (APLA), and new drugs such as low-molecular-weight heparin, which can offer better outcome in subsequent pregnancies. In addition, the chance to experience another pregnancy loss after two previous losses is around 30 % , and after three RPL, it is around 33 % [11, 23] . Thus, it might not be justifi ed to postpone clinical evaluation for the third pregnancy loss. This is especially true in view of the delay in childbearing age in most developed countries. An earlier evaluation may be further indicated if fetal cardiac activity was identifi ed before a loss, the woman is older than 35 years, or if the couple has had diffi culty conceiving [8] . Jaslow et al. [10] studied 1020 women with RPL and reported that the etiology was not statistically different between two, three, or more previous pregnancy losses. These authors recommended performing evaluation after two or more consecutive losses. In the present study, we focused on women with primary RPL and compared those with two RPL to those with three RPL with regard to the following aspects: etiology; demographic and obstetric characteristics; the course and outcome of the index pregnancy, including pregnancy complications, fetal death, and delivery outcomes; and complications for those who had live births. To our knowledge, these pregnancy and deli very outcomes have not been previously reported. We suggest that this information may provide the basis for appropriate and detailed consultations with the patients regarding etio logy, as well as the potential complications and outcome of pregnancy.
Material and methods
An RPL Clinic database was developed by the fi rst author (A.B.) using a computer program (Access 03; Microsoft, Redmond, WA, USA). All patients referred to this RPL Clinic in a University Teaching Hospital from 1996 to 2009 were included.
Study population
Women who were registered at the RPL Clinic in our University Medical Center during the study period 1996 – 2009, and who had two or more primary RPL according to the recent American Society for Reproductive Medicine guidelines were included in the study [1] . Ectopic pregnancy in pre-index pregnancies and multiple gestations were excluded from the study.
Statistical analysis
Univariate techniques were used to identify differences between the two groups. Student ’ s t -tests and χ 2 -tests were used to assess signifi cance for continuous and categorical variables, respectively. Data were analyzed using Statistical Package for the Social Sciences (SPSS) software (version 17.0; SPSS Inc., Chicago, IL, USA). The study protocol was approved by the Institutional Review Board (Helsinki Committee) of Soroka University Medical Center.
Data collection
Patient information entered onto a spreadsheet database included demographic and clinical characteristics such as parents ’ ages, occupation, ethnicity, chronic diseases and medications, obstetric history, and results of all evaluations for RPL. Information about the fi rst pregnancy following evaluation in the RPL Clinic, defi ned as the index pregnancy, was also collected. Data collection about further pregnancies occurring after the index pregnancy is ongoing. Information from medical records was abstracted and used to verify information obtained from the patient. All the information was reviewed independently by two physicians (A.B. and S.A.) to make sure that all the pregnancies were recorded and to confi rm the information. In the preconception visits to the clinic, medical and obstetric history was obtained and a physical examination was performed. Each patient was referred to a full examination for possible etiology for the RPL. The evaluation consisted of karyotypes of both partners; anatomic evaluation of the reproductive tract by hysteroscopy or three-dimensional (3D) ultrasound; hormonal evaluation by blood tests for prolactin, thyroid stimulating hormone (TSH), and fasting glucose; screening for autoimmune disease including tests for lupus anticoagulant activity [dilute Russell viper venom test and partial thromboplastin time-lupus anticoagulant (PTT-LA), confi rmed by a hexagonal phase phospholipid test] and immunoglobulins G (IgG) and M (IgM) anticardiolipin; and thrombophilic workup which included testing for protein C activity, protein S activity, antithrombin activity, activated protein C resistance, homocysteine level, factor V Leiden mutation, and factor II mutation. Test results were added to the patient charts.
For women who conceived following the evaluation, the standard protocol of the RPL Clinic included a transvaginal ultrasound examination using transducers 7 – 4 MHz to defi ne pregnancy location as well as fetal viability. Thereafter, they were seen in the same clinic on a regular basis every 3 – 4 weeks until delivery, in addition to the ambulatory regular follow-up.
Pregnancy was defi ned by ultrasound examination or histopathologic exam report. Pregnancy loss was defi ned as any spontaneous pregnancy loss occurring at or before 20 weeks of gestation or with a fetal weight ≤ 500 g including intrauterine fetal demise. Ectopic pregnancies and termination of pregnancy were counted in each patient ’ s total pregnancies, but were not considered spontaneous pregnancy losses. The miscarriages were divided into three groups: pre-embryonic loss (demise ≤ 6 weeks gestation), embryonic loss (demise ≥ 6 weeks gestation but < 10), and fetal loss (demise at ≥ 10 weeks but ≤ 20 weeks gestation).
Chronic diseases included diabetes mellitus, heart disease, hypertension, asthma, migraine, thromboembolism, epilepsy, genetic disease, and other diseases. Routine medications included any routine medications. Fertility problems were defi ned as failure to conceive during a period of 1 year or longer of regular contraceptive-free inter course. Past surgeries included appendectomy, cholecystectomy, gastric banding, gynecologic surgeries (including cesarean sections), and other surgeries.
Clinical laboratory investigation
Parental genetics – Signifi cant rearrangements (balanced translocations such as reciprocal or Robertsonian) were considered abnormal. Uterine anatomy – Uterine anatomic defects were identifi ed by hysteroscopy or 3D ultrasound. Considered abnormal were congenital uterine anomalies (such as septum, unicornuate, and bicornuate uteri), fi broids, polyps, as well as Asherman ’ s syndrome. Lupus anticoagulant – Serum levels of lupus anticoagulant were evaluated using the dilute Russell viper venom test and PTT-LA. Results > 42 s which were not corrected with a 1:1 mix with normal serum were considered abnormal if confi rmed by a hexagonal phase phospholipid test.
Anticardiolipin antibodies – Serum levels of anticardiolipin IgG and IgM were measured by enzyme-linked immuno assay; levels exceeding 15 U/mL were considered abnormal. All positive tests were confi rmed by repeat testing at least 12 weeks later.
Factor V Leiden mutation – This was considered abnormal if a heterozygous or homozygous factor V Leiden G1691A mutation was found.
Prothrombin (factor II) – Heterozygous or homozygous mutations for the G20210A prothrombin (factor II) gene were considered abnormal.
Thyroid function – Serum levels of TSH < 0.45 mIU/L or > 4.5 mIU/L were considered abnormal.
Blood glucose level – A patient ’ s serum glucose level was considered abnormal if fasting blood glucose was > 126 mg/dL.
Prolactin – Serum levels of prolactin which were > 23 ng/mL were considered abnormal.
Thrombophilic factors – Serum levels of functional protein C activity < 70 % , functional protein S activity < 70 % , or antithrombin activity < 75 % were considered abnormal. Fasting homocysteine – an abnormal result was recorded if > 17 mmol/L.
Minimal evaluation included parental karyotype, APLA eva luation, and anatomy evaluation.
Results
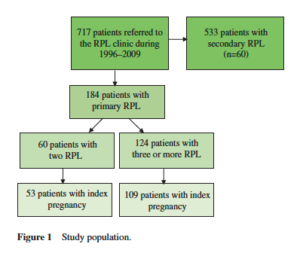
Of a total of 717 women who visited the RPL Clinic in Soroka University Medical Center during the study period 1996 – 2009, 184 had primary RPL. Sixty (32.6 % ) women had two consecutive pregnancy losses and 124 (67.4 % ) had three or more (Figure 1 ).
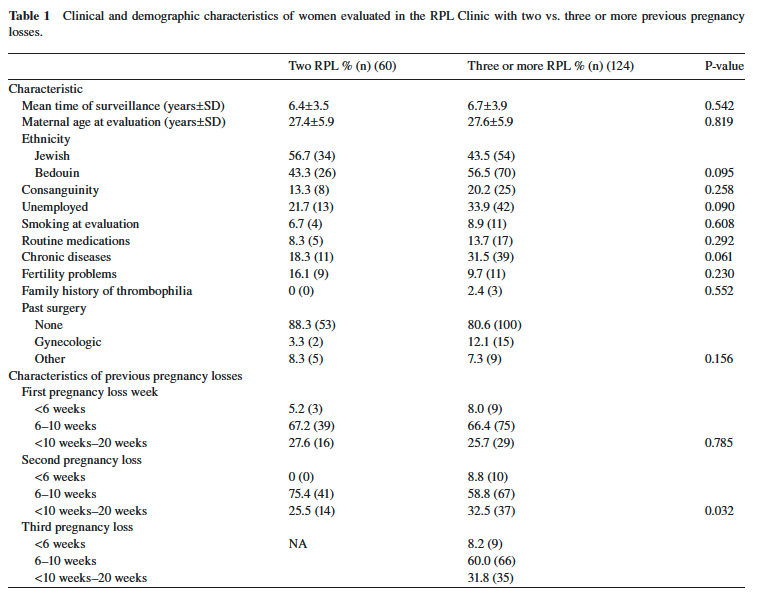
Table 1 contains information about maternal and obstetric characteristics in the two groups. All women were married, with no documented alcohol or drug use. The two groups were similar in most examined characteristics. In the three- RPL group compared to the two-RPL group, there were higher proportions of Bedouins, consanguineous marriages, unemployed women, and chronic diseases. However, none of these differences was statistically signifi cant. There was no difference in gestational age of the fi rst pregnancy loss, but there was a signifi cant difference between the two groups in the distribution of gestational age of the second pregnancy loss.
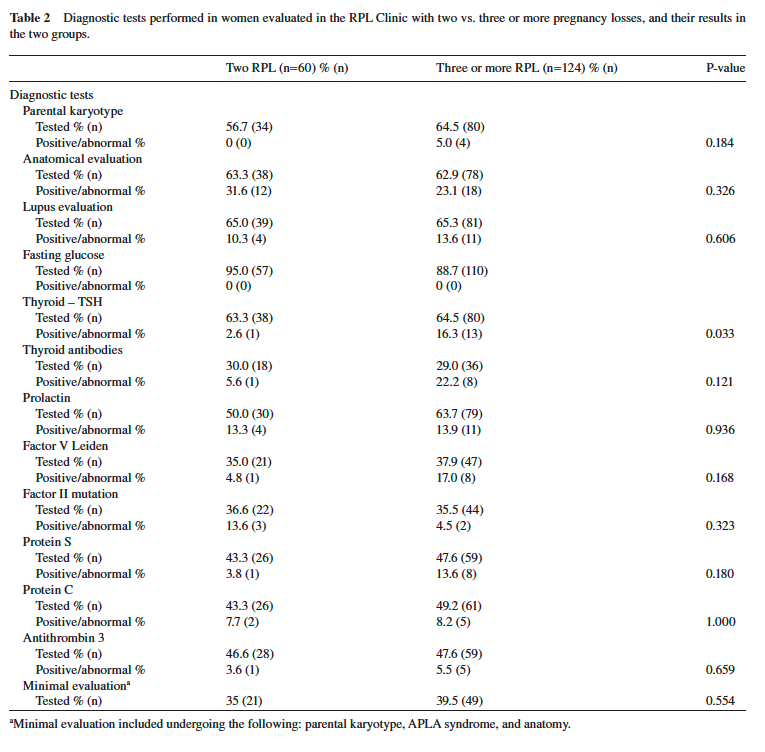
Table 2 shows the percentages of women in each study group who had undergone each diagnostic test and, among them, the percentage of positive or abnormal test results. In most tests performed, no statistically signifi cant difference was found in the rate of abnormal test results between the two groups, except for a signifi cantly higher rate of abnormal TSH levels. In women with three or more pregnancy losses, pregnancy occurred in 162 women (88 % ); 22 women had no record of pregnancy during the study period with no difference between the two groups. Of the pregnancies, 81.1 % resulted in births in the group of two pregnancy losses and 73.4 % of women with three or more pregnancy losses (P = 0.280).
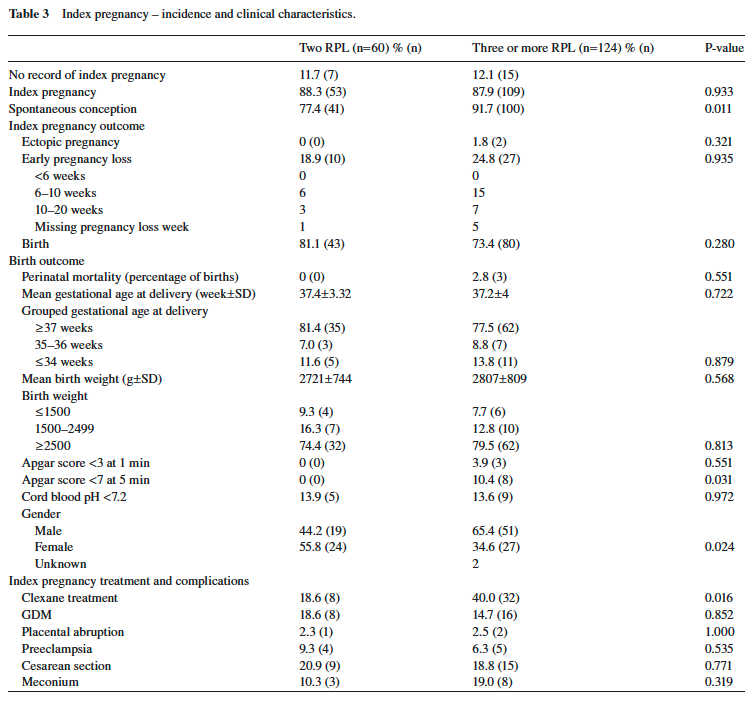
Low Apgar scores at 5 min were signifi cantly lower in neonates of women with two RPL compared to three or more RPL (0 vs. 10.8 % , respectively; P = 0.031). The rate of preterm delivery was lower in neonates of women with two RPL compared to three or more RPL (18.6% vs. 22.6 % , respectively) as was the rate of perinatal mortality (0% vs. 2.75 % , respectively). The differences, however, were not statistically signifi cant. A statistically signifi cant difference was found in infants ’ gender at the index pregnancy. Women with two pregnancy losses had a signifi cantly higher rate of female infants compared to women with three or more RPL (55.8% vs. 34.6 % , respectively; P = 0.024; Table 3 ).
No differences were found in rates of pregnancy loss and live birth between the two-RPL and three or more- RPL groups when stratifying on either spontaneous conception, or Clexane treatment (both variables on which the two study groups differed). When controlling for Clexane treatment, no difference was found in the rates of pregnancy complication presented in Table 3 (data not shown).
Discussion
This study supports recent changes in the defi nition of RPL and the need for evaluation after two consecutive pregnancy losses [10, 23] . The study included only those with primary RPL to examine a homogeneous group.
The proportion of women achieving pregnancy was similar in the two groups, although it was slightly higher in the two- RPL group than in the three-RPL group (81.1% vs. 73.4 % ), but the difference was not statistically signifi cant. This fi nding is in line with other studies [6, 20] .
The mean time of surveillance until the index pregnancy was 6.5 months, which was not different between the groups, and gave us the opportunity to give the couple tender loving care (TLC) and reduce stress, a well-known and accepted method in improving the live birth rate up to 80 % [6, 14, 20, 21] . In a very recent study which described patient attitude toward TLC, it was reported that among several aspects, the most important were early and frequently repeated ultrasounds, β-human chorionic gonadotropin monitoring, practical advice concerning lifestyle and diet, emotional support in the form of counseling, clear policy for the upcoming 12 weeks, and medication [16] .
The distribution of different etiologies was similar to that published in previous studies [2, 8, 17] , and in about 50 % of the patients, no etiology was found.
Most of the evaluation test results were similar in the two study groups except for the abnormal TSH level which was more common among the three-RPL women. Our findings are in agreement with Jaslow et al. [10] who investigated 1020 patients and did not find any differences in the evidence-based studies or in the investigational studies between women with two vs. three or more RPL. Thus, the similarity in test results among the two- RPL groups strongly suggested performing the evaluation after two RPL and not waiting for another pregnancy loss. A possible explanation for the abnormal TSH levels is the high level of thyroid antibodies which was four times higher in the three-RPL group, although it did not reach statistical significance. Several studies have been published recently on the significance of thyroid function, antibody, RPL, and the need for treatment [9, 18, 19, 25] . Thyroid replacement therapy appears to be more effective than intravenous immunoglobulin in preventing a new miscarriage [25] . A meta-analysis reported an association between the presence of thyroid autoantibodies, a history of one or two miscarriages, and the outcome of the next pregnancy. However, no association was found when analysis was restricted to those with RPL [18, 25] . The question of whether the thyroid antibody test is necessary at all or only in the three-RPL women is not clear. Further studies are needed to resolve this issue.
In this study, we examined several characteristics of the patients that, in part, were not previously reported, including consanguinity, employment, medication use, chronic disease, previous surgeries, fertility problems, and family history of thrombophilia. The only difference which was found between the groups was the chronic disease rate, which was higher in the three-RPL group (31.5% vs. 18.3 % ; P = 0.06). Our study population was not large enough to enable us to explore the specifi c chronic diseases which affected the patients. This issue needs to be further examined. In addition, several studies reported that RPL tends to affect women with specifi c reproductive characteristics such as conception delays/fertility investigations, late miscarriage (second trimester), stillbirth or neonatal death, ectopic pregnancy, abortion for fetal abnormality, previous live birth, pregnancy complicated by prematurity, or fetal growth restriction [2, 3, 7, 12, 18, 25] . This means that the RPL patients may be different from the normal population.
More RPL occurred after 10 weeks in the second pregnancies of the three-RPL group (32.5% vs. 25.5 % ; P = 0.032). Heuser et al. [9] reported on the gestational ages of women with RPL among idiopathic RPL and found no differences, but more information is needed to come to a clear conclusion.
With regard to the index pregnancy characteristics, the three-RPL group was more likely to be spontaneous in comparison to the two-RPL group (91.7% vs. 77.4 % ; P = 0.011), which might partially explain why the RPL couples were sent for evaluation after two RPL even before the change in the defi nitions. The fi nding refl ects the understanding that couples who have diffi culty conceiving require early intervention before another pregnancy loss occurs, especially when the couple does not have children, as none of their previous pregnancies resulted in a live birth.
Patients with three RPL were more likely to be treated with Clexane than women with two RPL. This difference was not related to thrombophilia alone but to empiric treatment which is a very common practice. Recent fi ndings indicate that Clexane treatment in idiopathic RPL does not change the live birth rate [5, 13] . Thus, we recently changed our policy according to these recent reports, and our protocol includes Clexane treatment in cases of thrombophilia – congenital or acquired – in patients with two or three RPL.
Additionally, a 5-min Apgar score of < 7 was more common in the three-RPL group (10.4% vs. 0 % ; P = 0.031). This may be due to the slightly higher rate of perinatal mortality in the three-RPL group.
Another fi nding in our study was a higher male rate in the three-RPL group (65.4% vs. 44.2 % ; P = 0.024). The theory about male abortus and the development of maternal immunity against male- specifi c minor histocompatibility (HY) antigens [4] does not support these fi ndings and needs further studies.
In summary, this study supports the approach which recommends evaluating couples with RPL after two consecutive pregnancy losses rather than three. The information presented in this study, including pregnancy complications and delivery outcomes, adds signifi cantly to the consideration that the two-RPL group needs evaluation as much as the three-RPL group.
Nowadays, child bearing in the western world is delayed, also justifying the changing defi nitions with recommendations to start evaluation at this point. The stress in those couples is high and they do not want to have another pregnancy loss and then start evaluation. As the rate of RPL is higher due to the new defi nition and in agreement with our results, early referral to the RPL Clinic means that more money is needed for the evaluation of this population.
It should be emphasized that performing evaluation after two consecutive RPL means that a higher rate of couples, between 2% and 5 % , will have to be included in this type of evaluation. Further studies are needed to assess the economic ramifi cations of the change in policy.
References
[1] American Society for Reproductive Medicine (ASRM). Defi nitions of infertility and recurrent pregnancy loss. Fertil Steril. 2008;89(Suppl 6):1603. Epub 2008 May 16.
[2] Branch DW, Gibson M, Silver RM. Clinical practice. Recurrent miscarriage. N Engl J Med. 2010;363(Suppl 18):1740 – 7.
[3] Brigham SA, Conlon C, Farquharson RG. A longitudinal study of pregnancy outcome following idiopathic recurrent miscarriage. Hum Reprod. 1999;14(Suppl 11):2868 – 71.
[4] Christiansen OB, Steffensen R, Nielsen HS. The impact of anti-HY responses on outcome in current and subsequent pregnancies of patients with RPL. J Reprod Immunol. 2010;85(Suppl 1): 9 – 14.
[5] Clark P, Walker ID, Langhorne P, Crichton L, Thomson A, Greaves M, et al. SPIN (Scottish Pregnancy Intervention) study: a multicenter, randomized controlled trial of low-molecularweight heparin and low-dose aspirin in women with recurrent miscarriage. Blood. 2010;115(Suppl 21):4162 – 7.
[6] Clifford K, Rai R, Regan L. Future pregnancy outcome in unexplained recurrent fi rst trimester miscarriage. Hum Reprod. 1997;12(Suppl 2):387 – 9.
[7] Clifford K, Rai R, Watson H, Regan L. An informative protocol for the investigation of recurrent miscarriage: preliminary experience of 500 consecutive cases. Hum Reprod. 1994;9:1328 – 32.
[8] Ford HB, Schust DJ. RPL: etiology, diagnosis, and therapy. Rev Obstet Gynecol. 2009;2(Suppl 2):76 – 83.
[9] Heuser C, Dalton J, Macpherson C, Branch DW, Porter TF, Silver RM. Idiopathic recurrent pregnancy loss recurs at similar gestational ages. Am J Obstet Gynecol. 2010;203(Suppl 4):343. e1 – e5.
[10] Jaslow CR, Carney JL, Kutteh WH. Diagnostic factors identifi ed in 1020 women with two versus three or more recurrent pregnancy losses. Fertil Steril. 2010;93(Suppl 4):1234 – 43. Epub 2009 Mar 31.
[11] Jauniaux E, Farquharson RG, Christiansen OB, Exalto N. Evidence-based guidelines for the investigation and medical treatment of RPL. Hum Reprod. 2006;21:2216 – 22.
[12] Jivraj S, Anstie B, Cheong YC, Fairlie FM, Laird SM, Li TC. Obstetric and neonatal outcome in women with a history of RPL: a cohort study. Hum Reprod. 2001;16(Suppl 1):102 – 6.
[13] Kaandorp SP, Goddijn M, van der Post JA, Hutten BA, Verhoeve HR, Hamulay á k K, et al. Aspirin plus heparin or aspirin alone in women with RPL. N Engl J Med. 2010;362(Suppl 17):1586 – 96.
[14] Liddell HS, Pattison NS, Zanderigo A. RPL – outcome after supportive care in early pregnancy. Aust N Z J Obstet Gynaecol. 1991;31(Suppl 4):320 – 2.
[15] Malpas P. A study of abortion sequence. J Obstet Gynecol Br Emp. 1938;45:932.
[16] Musters AM, Taminiau-Bloem EF, van den Boogaard E, van der Veen F, Goddijn M. Supportive care for women with unexplained RPL: patients ’ perspectives. Hum Reprod. 2011;26(Suppl 4): 873 – 7.
[17] Rai R, Regan L. Recurrent miscarriage. Lancet. 2006;368(Suppl 9535):601 – 11.
[18] Rushworth FH, Backos M, Rai R, Chilcott IT, Baxter N, Regan L. Prospective pregnancy outcome in untreated recurrent miscarriers with thyroid autoantibodies. Hum Reprod. 2000; 15(Suppl 7):1637 – 9.
[19] Stagnaro-Green A, Abalovich M, Alexander E, Azizi F, Mestman J, Negro R, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011;21(Suppl 10):1081 – 125.
[20] Stirrat GM. RPL. Lancet. 1990;336(Suppl 8716):673 – 5.
[21] Stray-Pedersen B, Stray-Pedersen S. Etiologic factors and subsequent reproductive performance in 195 couples with a prior history of habitual abortion. Am J Obstet Gynecol. 1984;148(Suppl 2): 140 – 6.
[22] Strobino B, Fox HE, Kline J, Stein Z, Susser M, Warburton D. Characteristics of women with recurrent spontaneous abortions and women with favorable reproductive histories. Am J Public Health. 1986;76(Suppl 8):986 – 91.
[23] The Dutch Society of Obstetrics and Gynecology (NVOG). Guideline RPL. The Netherlands: the Dutch Society of Obstetrics and Gynecology (NVOG); 2007 Aug 06. 20 pp.
[24] The investigation and treatment of couples with recurrent fi rsttrimester and second-trimester miscarriage. RCOG Green-top Guideline No. 17, April 2011; pp. 1 – 18.
[25] Vaquero E, Lazzarin N, De Carolis C, Valensise H, Moretti C, Ramanini C. Mild thyroid abnormalities and recurrent spontaneous abortion: diagnostic and therapeutical approach. Am J Reprod Immunol. 2000;43(Suppl 4):204 – 8.
The authors stated that there are no confl icts of interest regarding the publication of this article.
Received November 26, 2011. Revised January 11, 2012. Accepted January 19, 2012. Previously published online February 16, 2012.




